LACTIC ACID STUDY OF POST SLAUGHTERED ON PIGS IN ABBATOIR SANGGARAN DENPASAR BALI
on
Volume 14 No. 2: 148-153
April 2022
DOI: 10.24843/bulvet.2022.v14.i02.p11
Buletin Veteriner Udayana
pISSN: 2085-2495; eISSN: 2477-2712
Online pada: http://ojs.unud.ac.id/index.php/buletinvet
Terakreditasi Nasional Sinta 4, berdasarkan Keputusan Direktur Jenderal
Pendidikan Tinggi, Riset, dan Teknologi No. 158/E/KPT/2021
Lactic Acid Study of Post Slaughtered on Pigs in Denpasar Abbatoir Bali
(STUDI ASAM LAKTAT PASCA PEMOTONGAN PADA BABI DI RUMAH POTONG HEWAN DENPASAR BALI)
Siswanto1*, I Gede Soma1
1Laboratorium Fisiologi Veteriner, Fakultas Kedokteran Hewan, Universitas Udayana, Jl. PB. Sudirman Denpasar, Bali
*Email: siswantofkh@gmail.com
Abstract
The lactic acid in blood and meat can accelerate rigor mortis and reduce the quality of the meat, because the meat will rot quickly, thereby reducing its selling power. Increased levels of lactic acid in the blood / meat can be caused by improper cutting processes. This study aims to see the levels of lactic acid in the blood as monitoring. The experimental animals were used by 30 healthy pigs slaughtered at the slaughterhouse in Sanggaran, Denpasar, Bali. Sampling was done purposively. The blood sample was taken as much as 3 ml from the jugular vein. Elisa method is used for determination of plasma lactic acid levels. The results showed the range of lactic acid levels was 2.10 - 2.95 mmol / dL with a mean of 2.47 mmol / dL, Sd. 0.22. These profiles are in normal condition so it is not noticed that the handling or management before and during cutting is good.
Keywords: lactic acid, blood, pork.
Abstrak
Kadar asam laktat dalam darah dapat mempercepat rigor mortis dan mengurangi kualitas daging, karena daging akan cepat membusuk, sehingga akan menurunkan daya jualnya. Meningkatnya kadar asam laktat dalam darah/daging dapat disebabkan oleh proses pemotongan yang tidak baik. Penelitian ini bertujuan untuk mengetahui kadar asam laktat dalam darah sebagai monitoring. Hewan coba digunakan 30 ekor babi sehat yang disembelih di rumah potong hewan di Sanggaran, Denpasar, Bali. Pengambilan sampel dilakukan secara purposive. Sampel darah diambil sebanyak 3 ml dari vena jugularis. Metode elisa digunakan untuk penentuan kadar asam laktat plasma. Hasil menunjukkan range kadar asam laktat 2,10 – 2,95 mmol/dL dengan rata-rata 2.47 mmol/dL, Sd. 0.22. Profil tersebut berada pada kondisi normal sehingga disimpulkan bahwa penanganan atau menejemen sebelum dan saat pemotongan baik.
Kata kunci: asam laktat, darah, babi.
INTRODUCTION
Lactic acid meat can accelerate rigor mortis and the color of the meat becomes unattractive, so it will reduce the quality of the meat. Increased levels of lactic acid in the blood or meat can be caused by poor cutting management, therefore good handling is needed during cutting. The treatment during cutting will affect the levels of lactic acid which in turn will also affect the quality of the meat. Poor handling during cutting will increase lactic acid levels (aEdwards et al, 2010).
Tired animals and stressed have higher blood lactic acid levels. In a tired state,
animals lack energy, so the body oxidizes body fat deposits. Under anaerobic conditions, oxidation in cells converts pyruvate to lactate and produces low energy (ATP) (Kannan, et al, 2000). The plasma lactate level can be used as an indicator of anaerobic metabolism and lack of oxygen. When oxygen is available again (by providing a good ration), most of the lactate will be quickly converted to pyruvate and then enter the Krebs cycle, most of these changes occur in the liver (Gebresebet and Sällvik, 2006). Lactic acid will speed up the process of rigor mortis, and rigor mortis will reduce the quality of the meat. The
increase in lactic acid also results in a rapid decrease in the pH of the meat. Low pH of meat will result in a decrease in water binding capacity (DMA), thus affecting the level of chewiness of the meat (Borel, 2001; Aradom, 2013).
Lactic acid levels in the blood can be used to predict an animal's stress (Čobanović et al., 2020), lactate concentration affects carcass quality (aEdwards et al., 2010), bEdwards et al. (2010) handling animals before slaughter affects blood lactate concentrations (cEdwards et al., 2010).
Rigor mortis is an irreversible muscle stiffness, occurring after slaughter after primary relaxation. Rigor mortis occurs simultaneously in all voluntary and involuntary muscles. Rigor mortis in skeletal muscles occurs simultaneously in all muscles, but is usually more pronounced and easily observed in small muscles. The biochemical process that takes place before and after the animal is slaughtered until the formation of rigor mortis is generally a phase that plays a major role in the quality of the meat. Mishandling after slaughtering to the rigor mortis phase can result in low meat quality, characterized by dark (dark firm dry) or pale (pale soft exudative) meat or cold shortening or rigor that forms after melting of frozen meat (thaw rigor) (Dewi, 2012; ELkhasmi, 2013)
Excess carbohydrates from feed are converted into glycogen and stored in the liver and muscles. Furthermore, this glycogen will be overhauled into lactic acid (anaerobic) or pyruvic acid (aerobic) and produce ATP (adenosine tri phosphate) which is used for the process of muscle contraction and relaxation. After livestock die, the remains of glycogen and especially ATP formed during the pre-mortem are still used for muscle contraction until the ATP is depleted. As a result, rigor mortis will be formed, characterized by muscle stiffness. The production of ATP from glycogen goes through three pathways, namely the glycolysis pathway (the breakdown of glycogen into lactic acid or pyruvic acid).
Adenosine tri-phosphate (ATP) will be used as an energy source for contraction, pumping Ca2 ions during relaxation, and regulating the rate of Na and K equilibrium. The time required for rigor mortis to form depends on the amount of ATP available at the time of slaughter of cattle. The condition of livestock that does not rest before slaughter and especially in conditions of stress or fatigue will accelerate the formation of rigor mortis (Savenije et al., 2002).
Before rigor mortis occurs, the prarigor phase begins, where the muscles are still contracting and ending with the occurrence of stiffness in the muscles. The time required for rigor mortis to form depends on the amount of ATP available at the time the animal dies. The amount of ATP available depends on the amount of glycogen available at the time the animal dies. In animals that are tired / tired or stressed and lack of rest before being slaughtered, it will result in an insufficient supply of ATP so that the rigor mortis process will take place quickly. Likewise, high temperatures when animals are slaughtered will accelerate the depletion of ATP due to overhaul by the ATPase enzyme so that rigor mortis will take place quickly (de Castro, et al., 2017).
The circulatory system failure due to slaughter results in decreased oxygen supply in the muscles and eventually becomes depleted. According to Broom (2005), oxygen supply is cut off to the muscles due to slaughter due to stopping the work of the heart and blood circulation. The depleted oxygen supply causes energy metabolism or the breakdown of glycogen to lactic acid turns into anaerobic metabolism. The loss of blood results in the loss of temperature control in the muscles by the circulatory system. Heat from the inside of the body is no longer transported to the lungs and the surface of the body, resulting in an increase in temperature in the muscles and body after slaughter (Hofmaier et al, 2013).
The freshly slaughtered animal is flexible and soft, then changes occur where
the muscle tissue becomes hard, stiff and does not move easily. This situation takes a long time until it becomes soft again. The rigormortic phase of muscle tissue becomes hard and stiff, due to the depletion of ATP from the muscles. When the glycogen reserves run out, the formation of ATP will stop while the breakdown of ATP to produce energy continues, as a result, the amount of ATP in muscle tissue will gradually shrink. In the absence of ATP, there is no more energy that can maintain the function of the sarcoplasmic reticulin as a calcium pump, which is to keep the concentration of Ca ions around the myofilament as low as possible, resulting in the release of Ca ions. This Ca ionion will bind to the troponin protein, causing an electrostatic bond between actin and myosin filaments (actomyosin) and ultimately causing the meat to become hard and stiff (Hjyu, et al, 2002).
The short time for the formation of rigor mortis resulted in the pH of the meat was still high (above the normal pH of the meat) at the time of the formation of rigor mortis. If the pH is >5.5 – 5.8 when rigor mortis is formed with a faster time than normal conditions, the quality of the meat that will be produced will be low (dark red color, dry and tight structure) and will not last long in storage even at cold temperatures. In the rigor mortis phase, the glycolysis process will stop and the production of ATP will decrease, as a result anaerobic glycolysis occurs which produces lactic acid. As a result of reduced ATP, the distance between actin and myosin is getting closer which will eventually fuse to form actoniosin. This situation allows more contraction and causes the meat to stiffen. The next phase is the post rigor phase, in this phase the meat is contaminated with microbes, as a result, the meat becomes soft due to the role of the catepsin enzyme as a breakdown of actomyosin proteins into simple proteins (Broom, 2005).
RESEARCH METHODS
Research design
This observational study uses a crosssectional study design, with once independent variable being gender while the dependent variable is the levels of lactic acid.
Sample
Sampling was carried out using a purposive system (non-random or determined sampling), with 30 samples. The experimental animals used are healthy pigs that will be slaughtered at the Slaughterhouse, Pesanggaran, Denpasar, Bali. The sampling procedure is by taking approximately 3 ml of blood from the jugular vein. The collection was carried out at 03.00 WITA.
Approximately 3 ml of blood samples were taken from the jugular vein from 30 pigs inserted into a blood collecting tube (blood collecting tube) anticoagulated EDTA. It is put in a cooling box and taken to the laboratory to determine the level of lactic acid (indirect ELISA). The data obtained from Elisa were analyzed descriptively, tabulated and graphically.
Elisa method
Testing of lactic acid levels was carried out quantitatively using the Enzyme-linked Immunosorbent Assay (Elisa) method. Laboratory materials include 70% alcohol, cotton, and EDTA and elisa kit. The research instruments included a venojeck, test tube, cold flask and an elisa device.
Elisa procedure includes: Coating/capture–direct or indirect immobilization of antigens to the surface of polystyrene microplate wells. Plate blocking–addition of irrelevant protein or other molecule to cover all unsaturated surface-binding sites of the microplate wells. Probing/detection–incubation with antigen-specific antibodies that affinitybind to the antigens. Signal measurement– detection of the signal generated via the direct or secondary tag on the specific antibody.
Data analysis
The data is displayed in the range and average, standard deviation and presented in a graph, while the data analysis is done in a qualitative descriptive manner.
RESULTS AND DISCUSSION Results
The results of the ELISA test on lactic acid levels before the pigs were slaughtered from 30 blood samples are range 2,10 – 2,95 (mmol/dL), average 2.47 (mmol/dL), and SD 0.22.
Discussion
The range of blood lactic acid levels in Balinese pigs is 2.10 – 2.95 mmol/dL and an average of 2.47 mmol/dL, with an SD of 0.22. When compared to the research results of Hofmaier et al. (2013), 0.5 to 5.5 mmol/dL, Alstrup (2016), minipig (2.53 mmol/dL ± 1.10), showed that treatment during rest before slaughter was good. This shows that the handling during rest is done well such as sufficient time, adequate feed, comfortable environmental situation. As stated by Kannan et al. (2000) that
transportation involves several factors that can potentially cause stress to animals, including poor handling, mixing with foreign animals, age differences, lack of food and drink, transportation convenience, numbers and bad weather. Further, Grandin et al. (2000) explained that animals that are
not properly handled during transportation will become stressed. Stress can be observed by observing the clinical symptoms that appear. Animals that experience stress will experience a deficiency of certain metabolites such as blood glucose, electrolytes and water, so that the animal becomes dehydrated. Severe glycogen deficiency often occurs in conditions of fatigue, hunger, fear and heat stress or aggressive behavior due to mixing with foreign animals (Kannan et al., 2000). Stressed animals need recovery time, for example adequate rest, good feeding / drinking arrangements. During recovery the cows return to normal conditions (Al-Haidary, 2004; Borell, 2006; ELkhasmi, et al., 2013).
Lactate levels begin to increase when there is a need for energy by the body's muscles as a result of increased stress. Lack of oxygen causes lactic acid levels to increase, because lack of oxygen causes anaerobic metabolism in the body, so glucose is converted into lactic acid. Decreased glucose due to transportation stress results in increased mobilization of muscle glycogen for energy formation through the glycolysis process, thus causing low energy reserves (glycogen) in the liver and muscles, which in turn increases blood lactate levels (Aradom, 2013).
LACTIC ACID LEVELS
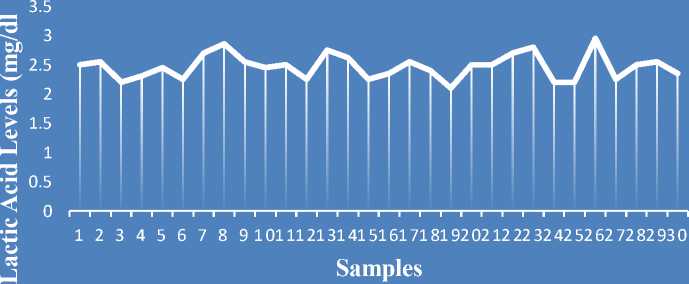
Figure 1. Graph of Pig Blood Lactic Acid Levels Before Slaughter
The most important pork quality traits (pH and temperature, water holding capacity, and color) had limited correlations with stress metabolites lactate acid (Čobanović et al., 2020). aEdwards, et al., (2010) stated that there was a relationship between blood lactate concentration and carcass quality in pigs. Furthermore, bEdwards, et al. 2010 argues that Pre-Slaughter Management of Pigs from Livestock to Processing Factory affects the Quality of Pork Meat.
CONCLUSION AND SUGGESTION
Conclusion
It was concluded that lactic acid levels showed normal, this means that the handling during slaughter was done well.
Suggestion
Needs handling before and on cutting is done.
ACKNOWLEDGEMENT
Thanks to the head of the abattoir, all staff and students who have helped so that this research is completed properly.
REFERENCES
Al-Haidary AA. 2004. Physiological
responses of naimey sheep to heat stress challenge under semi-arid environments. Int. J. Agric. Biol. 6(2): 307-309.
Alstrup AKO. 2016. Blood lactate concentrations in göttingen minipigs compared with domestic pigs. J. Am. Assoc. Lab. Anim. Sci. 55(1): 18-20.
Aradom S. 2013. Animal transport and welfare with special emphasis on transport time and vibration. Thesis. Uppsala: Swedish University of
Agricultural Sciences.
Borel EHV. 2001. The biology of stress and its application to livestock housing and transportation assessment. J. Anim. Sci. 79(Supl E): E260–E267.
Botha SSt, Hoffman, LC, Britz TJ. 2008.
Effects of post mortem temperature on rigor tension, shortening and pH in
ostrich muscle. SA. J. Anim. Sci. 38(3): 184.
Broom DM. 2005. The effect of land transport on animal welfare. Department of Veterinary Medicine. University of Cambridge. Madingley Road. Cambridge CB3 0ES.United Kingdom.
Čobanović N, Sanja Dj, Stanković SDj, Dimitrijević M., Suvajdžić B, Grković N, Vasilev D, Karabasil N. 2020. Identifying physiological stress biomarkers for prediction of pork quality variation. Animals. 10(4): 614. Doi: 10.3390/ani10040614
de Castro PL, Lewandowski V, de Souza MLR, Coradini MF, Alexandre AAC, César Sary, C, Ribeiro RP. 2017. Effect of different periods of pre-slaughter stress on the quality of the nile tilapia meat. Food Sci. Technol. 37(1): 52-58.
Dewi SHC. 2012. Korelasi antara kadar glikogen, asam laktat, pH daging dan susut masak daging domba setelah pengangkutan. J. Agri. Sains. 4(5): 5970.
aEdwards LN, Engle TE, Correa JA, Paradis MA, Grandin T, Anderson DB. 2010. The relationship between
exsanguination blood lactate
concentration and carcass quality in slaughter pigs. Meat Sci. 85(3): 435440. doi:
10.1016/j.meatsci.2010.02.012.
bEdwards LN, Grandin T, Engle TE, Ritter MJ, Sosnicki AA, Carlson BA, Anderson DB. 2010. The effects of preslaughter pig management from the farm to the processing plant on pork quality. Meat Sci. 86(4): 938-944. doi: 10.1016/j.meatsci.2010.07.020.
cEdwards LN, Grandin T, Engle TE, Porter SP, Ritter MJ, Sosnicki AA, Anderson DB. 2010. Use of exsanguination blood lactate to assess the quality of preslaughter pig handling. Meat Sci. 86(2): 384-390. doi:
10.1016/j.meatsci.2010.05.022.
ELkhasmi M, Chakir Y, Riad F, Safwate A, Tahri EH, Farh M, El Abbadi N,
Abouhafs R, Faye B. 2013. Effect of transportation stress during the hot-season on some haematological and physiological parameters in moroccan camels (Camelus dromedarius). J. Life Sci. 7(1): 13-25.
Gebresebet G, Sällvik K. 2006. Animal Welfare Aspects -the Technical Challenge During Housing, Transport and Slaughter: Dairy Cows.
Grandin T. 2000. Livestock Handling and Transport. 2th Ed. Departement of
Animal Science, Colorado University. ISBN: 0-85199-409-1. USA: Cabi
publishing.
Hofmaier F, Dinger K, Braun R, Sterner-Kock A. 2013. Range of blood lactate values in farm pigs prior to experimental surgery. Lab. Anim. 47(2):130-132. doi:
10.1177/0023677213476857.
Hjyu EJ, Pösö AR, Ruusunen MH,
Sepponen KV, Kylä-Puhju MS. 2002. Lactic acid in muscle and its effects on meat quality. Proc. The 55th reciprocal meat conference. Department of Food Technology/Meat Technology
University of Helsinki Viikki E, P.O. Box 27, FIN - 00014 Helsinki, Finland.
Kannan G, Terrill TH, Kouakou B, Gazal OS, Gelaye S, Amoah EA, Samake S. 2000. Transportation of goats: Effects on physiological stress responses and live weight loss. J. Anim. Sci. 78(6): 1450-1457.
Savenije B, Lambooij E, Gerritzen MA, Venema K, Korf J. 2002. Effects of feed deprivation and transport on preslaughter blood metabolites, early postmortem muscle metabolites, and meat quality. Poult. Sci. 81(5): 699-708.
153
Discussion and feedback